JAKAFI® or JAKAFI XR™ (ruxolitinib) is a prescription medication used to treat adults with certain types of myelofibrosis (MF). It is not known if JAKAFI or JAKAFI XR is safe or effective in children for treatment of myelofibrosis.
Before you start taking JAKAFI® (ruxolitinib) or JAKAFI XR™ (ruxolitinib) and throughout the duration of your treatment, your doctor will perform a blood test called a complete blood count (CBC). This will help them:
Each person with myelofibrosis (MF) is unique, and your doctor will determine the optimal dosage strength of JAKAFI or JAKAFI XR that’s right for you. While your response to JAKAFI or JAKAFI XR is being monitored, it may be necessary to increase or decrease your dose, or to pause your treatment. This is a conversation that you should have with your doctor.
Because everyone is different, and because your dosage may change during treatment, JAKAFI and JAKAFI XR are available in different dosage strengths. You should take the dose prescribed to you by your doctor.

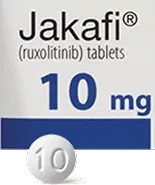
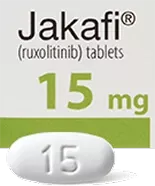


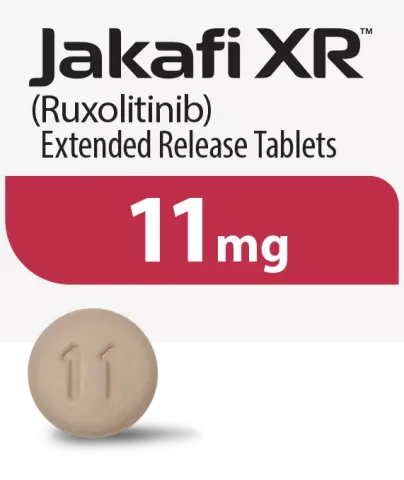




Tablets shown are not actual size.
For illustration purposes only.
Your doctor will determine the best dosing option for you by taking several factors into account, including:
For intermediate or high-risk MF, the recommended starting dose is based on your platelet counts. Before you start JAKAFI or JAKAFI XR, your doctor will perform a complete blood count to determine the appropriate dose.
During treatment, your doctor may check your blood cell counts regularly to make sure they don’t become too low. Depending on how you respond to JAKAFI or JAKAFI XR, your doctor may increase or decrease the dose, or pause treatment, based on your blood counts and symptoms. In some instances, your doctor may have to temporarily stop your treatment with JAKAFI or JAKAFI XR.
Always follow your doctor’s directions. It’s important to take JAKAFI or JAKAFI XR exactly as your doctor prescribes. Take JAKAFI XR exactly as directed; swallow it whole. Do not crush, break, or chew.
Take JAKAFI or JAKAFI XR at home or on the go. JAKAFI or JAKAFI XR is a prescription medicine available as a tablet that’s taken orally when it fits into your routine.
JAKAFI or JAKAFI XR can be taken with or without food.
Take JAKAFI or JAKAFI XR around the same time every day. Try taking it at the same time as you perform another daily activity, like brushing your teeth.
If you miss a dose of JAKAFI or JAKAFI XR, take your next dose at your regular time. Do not take 2 doses at the same time. If you take too much JAKAFI or JAKAFI XR, call your doctor or go to the nearest hospital emergency room right away. Take the bottle of JAKAFI or JAKAFI XR with you.
Before taking JAKAFI or JAKAFI XR, tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, or herbal supplements you’re taking. Some medicines can change how JAKAFI or JAKAFI XR works, so it’s important to share everything with your doctor. Be sure to mention any health problems you may have or had in the past, if you are pregnant, planning to get pregnant, or breastfeeding.
For more information about getting your JAKAFI or JAKAFI XR prescription filled, contact IncyteCARES for JAKAFI at 1-855-452-5234 or visit IncyteCARES.com/JAKAFI.

Register for the Incyte Mentor Program and connect with another patient taking Jakafi.
Learn How
Discover how Sue’s MF symptoms prompted her to talk to her Healthcare Professional about moving her treatment journey forward with Jakafi.
Watch Sue’s StoryJAKAFI or JAKAFI XR (ruxolitinib) is a prescription medication used to treat adults with certain types of myelofibrosis (MF).
It is not known if JAKAFI or JAKAFI XR is safe or effective in children for treatment of myelofibrosis.
JAKAFI or JAKAFI XR is a prescription medication used to treat adults with polycythemia vera (PV) who have already taken a medicine called hydroxyurea and it did not work well enough or they could not tolerate it.
It is not known if JAKAFI or JAKAFI XR is safe or effective in children for treatment of polycythemia vera.
JAKAFI or JAKAFI XR is a prescription medication used to treat adults and children 12 years of age and older with acute graft-versus-host disease (GVHD) who have taken corticosteroids and they did not work well enough.
JAKAFI or JAKAFI XR is a prescription medication used to treat adults and children 12 years of age and older with chronic graft-versus-host disease (GVHD) who have taken one or two types of treatments and they did not work well enough.
JAKAFI or JAKAFI XR can cause serious side effects, including:
Low blood counts: JAKAFI or JAKAFI XR may cause low platelet, red blood cell, and white blood cell counts. If you develop bleeding, stop taking JAKAFI or JAKAFI XR and call your healthcare provider. Your healthcare provider will do a blood test to check your blood counts before you start and regularly during your treatment. Your healthcare provider may change your dose or stop your treatment based on the results of your blood tests.
Tell your healthcare provider right away if you develop or have worsening symptoms such as
Infection: You may be at risk for developing a serious infection during treatment with JAKAFI or JAKAFI XR. Tell your healthcare provider if you develop any of the following symptoms of infection:
Worsening of symptoms after interrupting or stopping treatment. Signs and symptoms of myelofibrosis may worsen after you stop treatment.
Do not interrupt or stop treatment without talking to your healthcare provider. Tell your healthcare provider right away if you have any of the following after stopping treatment:
Cancer: Some people have had certain types of non-melanoma skin cancers during treatment with JAKAFI or JAKAFI XR. Your healthcare provider will regularly check your skin during your treatment. Tell your healthcare provider if you develop any new or changing skin lesions during treatment.
Increases in cholesterol: You may have changes in your blood cholesterol levels during treatment with JAKAFI or JAKAFI XR. Your healthcare provider will do blood tests to check your cholesterol levels about every 8 to 12 weeks after you start taking JAKAFI or JAKAFI XR and as needed.
Increased risk of major cardiovascular events such as heart attack, stroke or death in people who have cardiovascular risk factors and who are current or past smokers while using another JAK inhibitor to treat rheumatoid arthritis:
Get emergency help right away if you get any symptoms of a heart attack or stroke during treatment with JAKAFI or JAKAFI XR, including:
Increased risk of blood clots: Blood clots in the veins of your legs (deep vein thrombosis, DVT) or lungs (pulmonary embolism, PE) have happened in people taking another JAK inhibitor for rheumatoid arthritis and may be life-threatening.
Tell your healthcare provider right away if you have any signs and symptoms of blood clots during treatment with JAKAFI or JAKAFI XR, including:
Possible increased risk of new (secondary) cancers: People who take another JAK inhibitor for rheumatoid arthritis have an increased risk of new (secondary) cancers, including lymphoma and other cancers. People who smoke or who smoked in the past have an added risk of new cancers.
The most common side effects of JAKAFI or JAKAFI XR include:
These are not all the possible side effects. Ask your pharmacist or healthcare provider for more information. Call your doctor for medical advice about side effects.
Before taking JAKAFI or JAKAFI XR, tell your healthcare provider about:
Women should not take JAKAFI or JAKAFI XR while pregnant or planning to become pregnant. Do not breastfeed during treatment with JAKAFI or JAKAFI XR and for 2 weeks after the final dose.
How should I take JAKAFI or JAKAFI XR?
Please see the Full Prescribing Information, including Patient Information, which includes a more complete discussion of the risks associated with JAKAFI or JAKAFI XR.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088.
You may also report side effects to Incyte Medical Information at 1-855-463-3463.