About Acute Graft-Versus-Host Disease
What is acute graft-versus-host disease?
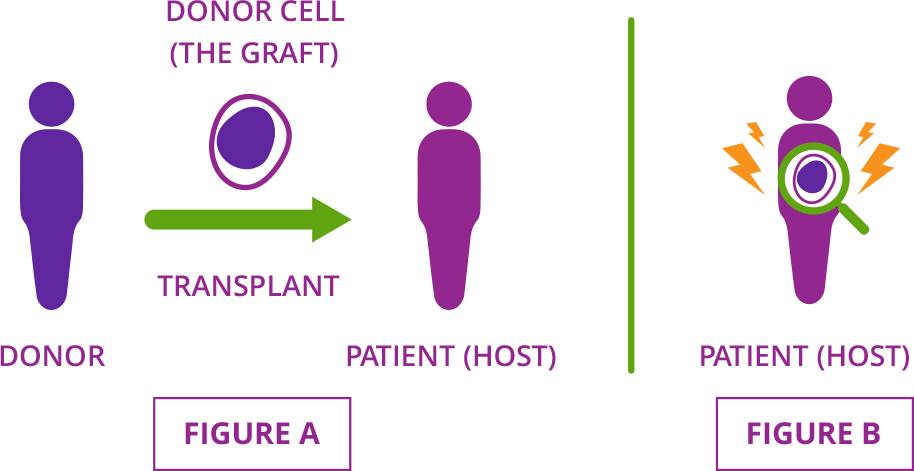
Acute graft-versus-host disease (GVHD) is a serious complication that may affect people who have had a stem cell transplant using cells from a donor. This type of procedure is called an allogeneic (AL-oh-geh-NAY-ik) stem cell transplant. During an allogeneic stem cell transplant, a patient’s cells are replaced with donor cells. (Figure A)
Acute GVHD occurs when donor cells (called the graft) attack the organs and tissues of the patient who received them (or the host). That’s why the condition is known as graft-versus-host disease. (Figure B)
Although acute GVHD can occur any time after transplant, it is commonly diagnosed within the first few months. Your Transplant Team relies on a combination of factors—including your symptoms and what organs are affected—to diagnose acute GVHD.
What parts of the body are affected by acute GVHD?
Acute GVHD mainly affects the skin, liver, and gastrointestinal tract (stomach, intestines, and colon). Patients may experience symptoms or other health problems in any of these organs because of acute GVHD.
What are the symptoms and signs of acute GVHD?
The following are all common symptoms and signs of acute GVHD. If you or a loved one notice that you are experiencing any new or worsening symptoms, contact your Transplant Team right away. Taking action early with acute GVHD can make a difference in your recovery and health.

Skin
Faint to severe sunburn-like rash (can appear anywhere, may cover entire body) | Blistering and peeling skin

Gastrointestinal tract
(stomach, intestines)
Diarrhea | Abdominal pain | Nausea | Vomiting | Bloating | Blood in the stool

Liver
Effects identified by blood tests | Few outward signs, but may include: yellowing of skin or eyes (jaundice); pain in the upper part of your belly; dark, tea-colored urine
What is acute GVHD “grading”?
Once your Transplant Team diagnoses acute GVHD, they will also give it a grade. The grade reflects the severity of acute GVHD and is based on the number of organs involved and how advanced symptoms are in each one. The grade is then used to guide treatment decisions and to help your team monitor for changes in your condition.
Transplant centers sometimes use different grading systems, so it’s a good idea to ask how the severity of your acute GVHD is being measured.

Already taking Jakafi? IncyteCARES for Jakafi is a patient support program helping eligible patients with access and support.
Learn More
Hear From Incyte’s Experienced Nurse Educators.
In this video series, our team shares their insights on common acute GVHD questions and questions related to Jakafi.
View VideosWhat is JAKAFI® or JAKAFI XR™?
What is JAKAFI® or JAKAFI XR™?
JAKAFI or JAKAFI XR (ruxolitinib) is a prescription medication used to treat adults with certain types of myelofibrosis (MF).
It is not known if JAKAFI or JAKAFI XR is safe or effective in children for treatment of myelofibrosis.
JAKAFI or JAKAFI XR is a prescription medication used to treat adults with polycythemia vera (PV) who have already taken a medicine called hydroxyurea and it did not work well enough or they could not tolerate it.
It is not known if JAKAFI or JAKAFI XR is safe or effective in children for treatment of polycythemia vera.
JAKAFI or JAKAFI XR is a prescription medication used to treat adults and children 12 years of age and older with acute graft-versus-host disease (GVHD) who have taken corticosteroids and they did not work well enough.
JAKAFI or JAKAFI XR is a prescription medication used to treat adults and children 12 years of age and older with chronic graft-versus-host disease (GVHD) who have taken one or two types of treatments and they did not work well enough.
Important Safety Information
JAKAFI or JAKAFI XR can cause serious side effects, including:
Low blood counts: JAKAFI or JAKAFI XR may cause low platelet, red blood cell, and white blood cell counts. If you develop bleeding, stop taking JAKAFI or JAKAFI XR and call your healthcare provider. Your healthcare provider will do a blood test to check your blood counts before you start and regularly during your treatment. Your healthcare provider may change your dose or stop your treatment based on the results of your blood tests.
Tell your healthcare provider right away if you develop or have worsening symptoms such as
- unusual bleeding
- bruising
- tiredness
- shortness of breath
- a fever
Infection: You may be at risk for developing a serious infection during treatment with JAKAFI or JAKAFI XR. Tell your healthcare provider if you develop any of the following symptoms of infection:
- chills
- nausea
- vomiting
- aches
- weakness
- fever
- painful skin rash or blisters
Worsening of symptoms after interrupting or stopping treatment. Signs and symptoms of myelofibrosis may worsen after you stop treatment.
Do not interrupt or stop treatment without talking to your healthcare provider. Tell your healthcare provider right away if you have any of the following after stopping treatment:
- fever
- trouble breathing
- weakness
- night sweats
- feeling dizzy or lightheaded
- pain in left upper stomach area or left shoulder
Cancer: Some people have had certain types of non-melanoma skin cancers during treatment with JAKAFI or JAKAFI XR. Your healthcare provider will regularly check your skin during your treatment. Tell your healthcare provider if you develop any new or changing skin lesions during treatment.
Increases in cholesterol: You may have changes in your blood cholesterol levels during treatment with JAKAFI or JAKAFI XR. Your healthcare provider will do blood tests to check your cholesterol levels about every 8 to 12 weeks after you start taking JAKAFI or JAKAFI XR and as needed.
Increased risk of major cardiovascular events such as heart attack, stroke or death in people who have cardiovascular risk factors and who are current or past smokers while using another JAK inhibitor to treat rheumatoid arthritis:
Get emergency help right away if you get any symptoms of a heart attack or stroke during treatment with JAKAFI or JAKAFI XR, including:
- discomfort in the center of your chest that lasts for more than a few minutes or that goes away and comes back
- severe tightness, pain, pressure, or heaviness in your chest, throat, neck, or jaw
- pain or discomfort in your arms, back, neck, jaw, or stomach
- shortness of breath with or without chest discomfort
- breaking out in a cold sweat
- nausea or vomiting
- feeling lightheaded
- weakness in one part or on one side of your body
- slurred speech
Increased risk of blood clots: Blood clots in the veins of your legs (deep vein thrombosis, DVT) or lungs (pulmonary embolism, PE) have happened in people taking another JAK inhibitor for rheumatoid arthritis and may be life-threatening.
Tell your healthcare provider right away if you have any signs and symptoms of blood clots during treatment with JAKAFI or JAKAFI XR, including:
- swelling, pain, or tenderness in one or both legs
- sudden, unexplained chest or upper back pain
- shortness of breath or difficulty breathing
Possible increased risk of new (secondary) cancers: People who take another JAK inhibitor for rheumatoid arthritis have an increased risk of new (secondary) cancers, including lymphoma and other cancers. People who smoke or who smoked in the past have an added risk of new cancers.
The most common side effects of JAKAFI or JAKAFI XR include:
- for certain types of myelofibrosis (MF) – low platelet or red blood cell counts, bruising, dizziness, headache, and diarrhea
- for certain types of polycythemia vera (PV) – low platelet or red blood cell counts, bruising, dizziness, headache, and diarrhea
- for acute GVHD – low platelet counts, low red or white blood cell counts, infections, and swelling
- for chronic GVHD – low red blood cell or platelet counts and infections, including viral infections
These are not all the possible side effects. Ask your pharmacist or healthcare provider for more information. Call your doctor for medical advice about side effects.
Before taking JAKAFI or JAKAFI XR, tell your healthcare provider about:
- all the medications, vitamins, and herbal supplements you are taking
- your medical conditions, including if you
- have or had low white or red blood cell counts
- have an infection
- have or had tuberculosis (TB) or have been in close contact with someone who has TB
- had shingles (herpes zoster)
- have or had hepatitis B
- have a high level of fat in your blood (high blood cholesterol or triglycerides)
- have or have had a blood clot, heart attack, other heart problems, or stroke
- are a current or past smoker
- had cancer
- have or had liver or kidney problems or are on dialysis. If you are on dialysis, JAKAFI or JAKAFI XR should be taken after your dialysis
- have any other medical condition
Women should not take JAKAFI or JAKAFI XR while pregnant or planning to become pregnant. Do not breastfeed during treatment with JAKAFI or JAKAFI XR and for 2 weeks after the final dose.
How should I take JAKAFI or JAKAFI XR?
- Take exactly as your healthcare provider tells you.
- Do not change your dose or stop taking JAKAFI or JAKAFI XR without first talking to your healthcare provider. If you stop treatment, symptoms of your condition may return.
- If you miss a dose, take your next dose at your regular time. Do NOT take 2 doses at the same time.
- You may have regular blood tests during your treatment. Based on the results, your healthcare provider may change your dose or stop JAKAFI or JAKAFI XR.
- IF YOU ARE PRESCRIBED JAKAFI:
- Take JAKAFI 2 times a day with or without food.
- JAKAFI may also be given through certain nasogastric tubes.
- Tell your healthcare provider if you cannot take JAKAFI by mouth. Your healthcare provider will decide if you can take JAKAFI through a nasogastric tube. Ask your healthcare provider to give you specific instructions on how to properly take JAKAFI through a nasogastric tube.
- IF YOU ARE PRESCRIBED JAKAFI XR:
- Take JAKAFI XR 1 time a day with or without food.
- Swallow JAKAFI XR whole. Do not split, crush, or chew.
Please see the Full Prescribing Information, including Patient Information, which includes a more complete discussion of the risks associated with JAKAFI or JAKAFI XR.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088.
You may also report side effects to Incyte Medical Information at 1-855-463-3463.



